Translate this page into:
Horse-Shoe Flap Reconstruction Technique in Breast Surgery: From The Smallest Defect to The Biggest—A Case Series
Address for correspondence Ida Bagus Made Suryawisesa, PhD, Division of Oncology Surgery, General Surgery Department, Faculty of Medicine Udayana University, Sanglah General Hospital, Jl. Diponegoro, Dauh Puri Klod, West Denpasar, Denpasar, Bali, Indonesia, 80113gusdeck2001@yahoo.com
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Chest wall resections and reconstructions are routinely performed to close defects after primary breast tumor resections. Depending on the size of primary breast tumors, the reconstructive technique requires more challenging approaches. In this study, we would like to introduce a novel technique which can be applied as a method in covering various sizes of skin defects after mastectomy termed as “horse-shoe flap technique.” We conducted a series of case reports of patients who underwent chest wall coverage after primary breast tumor resections between October 2018 and November 2019 and selected five cases of breast cancer patients. These patients were treated with the horse-shoe flap technique which relies on shape as its main concept. All chest defects must form a circular shape, based on the principle that adequate safe tumor margins are best achieved with a circular incision. The donor flap was drawn and taken from the outer circle area with a larger arc, depending on the defect's diameter size. Five post-mastectomy chest wall defect cases had been successfully closed with this technique. Patients were all female, ranging from 36 to 55 years of age. The smallest chest defect was 4 cm in diameter, and the largest was 26 cm. All the defects were closed using a 120-degree horse-shoe flap design. One patient experienced marginal necrotic skin, and one case had formed postoperative seroma. The horse-shoe flap technique proves to be an excellent option for closing chest skin defects which can be applied for various sizes of skin defects with minor complications.
Keywords
horse-shoe flap
breast cancer
breast reconstruction
Introduction
Surgical science has improved for the past decades, allowing the development and refinement of reconstruction techniques, particularly in chest wall areas. Along with improvements in many surgical fields, such as anesthesia and critical care medicine, chest wall reconstructions can be performed with decreased morbidity, mortality, and better outcomes.1 Chest wall resections and reconstructions are routinely performed nowadays; the most common reasons are primary breast tumors. Some chest wall resections leave large skin defects, depending on how large the size of primary breast tumors, and could result in more difficult reconstructive approaches.2 Nowadays, the large skin defects secondary to large primary breast cancer tumors had proven to be challenging problems for surgeons.
There are numerous reconstruction techniques available which have previously been conducted to cover chess wall defects. These reconstruction techniques are generally divided into two main categories: soft tissue reconstructions with or without prosthetic materials.3 Chest wall soft tissue reconstructions, such as latissimus dorsi (LD) muscle flap, pectoralis major muscle flap, serratus anterior muscle flap, and rectus abdominis muscle flaps were commonly used for chest skin defect coverage; however, some techniques cause complications.4
In this literature, we would like to introduce a new technique which can be used as a method in covering a large skin defect. It has a perfect perforator supply due to its design which resulted in minimal damage and defect to the donor site and more importantly, it is easy to perform. We will now introduce our novel design flaps reconstruction for big circular defect: a “horse-shoe flap technique” which has been applied particularly in chest wall reconstruction cases in our center. This method perfectly achieved a minimal-tension wound edge which ensured dermal perfusion. In this study, we reported several post-mastectomy with large chest skin defects coverage using our new horse-shoe flap technique. This report aims to introduce a new approach in covering skin defects secondary to mastectomy.
Methods
In this study, we conducted a series of case reports of patients who underwent chest wall soft tissue coverage after mastectomy or breast-conserving therapy due to breast malignancies or primary breast tumors. The duration of this retrospective study is 1 year, from October 2018 to November 2019. All of the operations and chest wall reconstructions were performed by our oncology surgeons in Sanglah General Hospital in Denpasar, Bali. Sanglah General Hospital, located in West Denpasar, is the biggest referral hospital in Bali, receives numerous patients from all around Bali, Nusa Tenggara Islands, and other areas in East Java, particularly breast cancer patients for curative and palliative approaches. Sanglah General Hospital is also the main teaching hospital for Udayana University, Faculty of Medicine, including our Department of Oncology Surgery. All patients included in this study were informed and have provided their consent to be included in the study.
We selected five representative cases of breast cancer patients with various sizes of primary breast tumors. These patients were all candidates for radical mastectomy and breast reconstruction. Our study sample's inclusion criteria are patients who underwent breast conserving operation and mastectomy which left a decent size of soft tissue defects. There are no limitations in tumor or defect size, as we would like to prove the availability of this technique which can be applied for the size of every defect, as long as the adjacent skin length would suffice for coverage. Patients with tumors or operative approaches affecting the surrounding thoracic structure, such as ribs, sternum, and clavicles, were excluded from our study samples. All clinical variables were recorded and presented, including age, diagnosis of breast diseases, methods, defects size, defects location, flap design and length of adjacent skin needed for closure.
Horse-Shoe Flap Technique
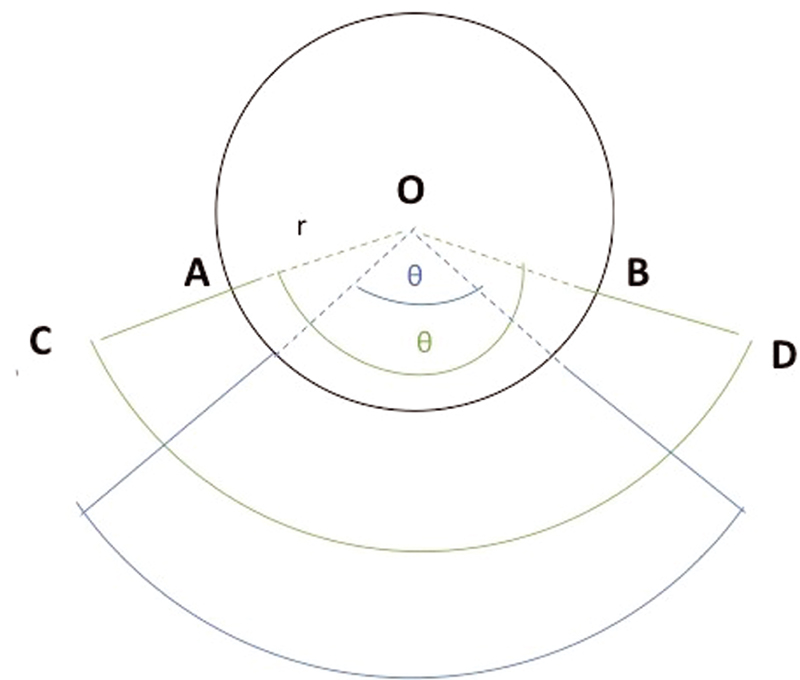
The horse-shoe flap is one of the reconstruction techniques that rely on shape as its main concept. Shape reconstruction techniques are basically easier to perform but this shape-concept technique appears to be abandoned as surgeons prefer to use the anatomy-based flap. For this horse-shoe flap technique, all chest defects must be formed into a circular shape defect a circular shape. This is also based on the principle that adequate safe tumor margins can be achieved with the use of a circular incision.5 For that reason, before the wide excision or mastectomy is performed, a circular pattern must be drawn and note the length of defects diameter, as it will determine the length of the adjacent donor flap. The donor flap is drawn and taken from the outer circle area with a larger arc, depending on the defects diameter size. A mathematical formula is created to determine the length of secondary defect size or donor area, which needs to be dissected (Fig. 1). Prior to surgery, the dimension of the potential defect after tumor ablation is estimated and outlined.
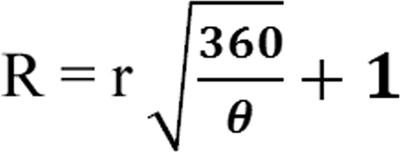
R: Donor flap length (in cm) or radius of the outer circle area.
r: Radius of the circular defect (in cm).
θ: Central circle angle.
-
Fig. 1 The horse-shoe flap's concept with circular shape pattern. Note the angle from the center circle and the defect's radius, which will determine the size of adjacent flaps needed for closure.
To facilitate determining the length of the donor flap, there are three basic choices of central circle angle and the length of donor flap needed for closure. The three central circle angles mentioned before are 180, 120, and 90 degrees. If we use 180 degrees as our circular center angle, we only need 73% of defect radius as donor flap length. If we used 120 degrees as our circular center angle, we need the length of defect radius as donor flap length.
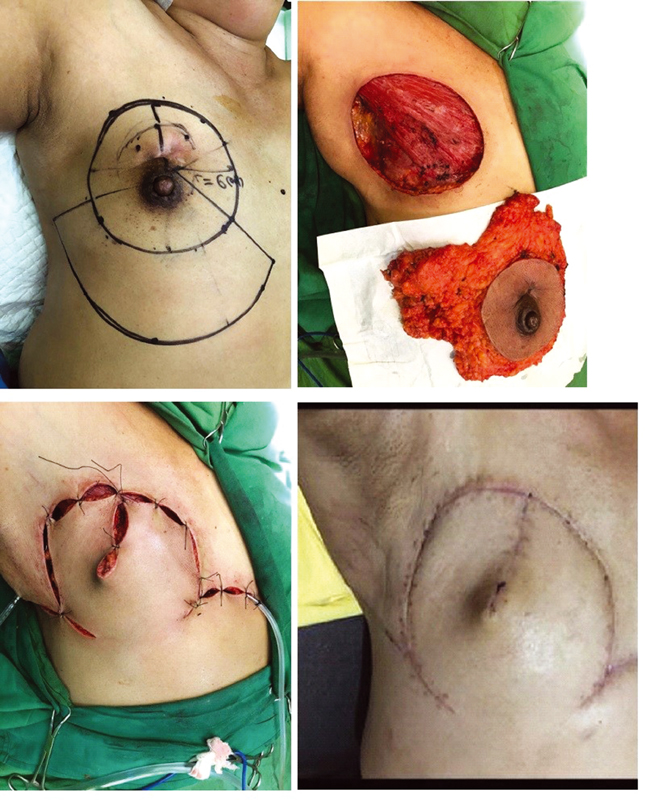
For the closure, both ends of the larger arc of the keystone flap are closed by Y suture, leaving only a small defect in the middle part. The end of the donor flap that meets at the center of the defect will be stitched in a straight line in the middle to obtain a good cosmetic reconstruction (Fig. 2). This flap viability depends on its perforator vessels, taken together with adjacent neural components to improve flap's perfusion, making this horse-shoe flap defined as one of the musculocutaneous flaps.
-
Fig. 2 Surgical steps on performing horse-shoe flap using outer circle adjacent tissue as donor flap.
Results
Five post-mastectomy chest wall defect cases were successfully closed with our new “horse-shoe flap techniques.” Our study samples were all female patients aged between 36 to 55 years of age; the smallest chest defect was measured 4 cm in diameter and the largest was 26 cm. One patient had marginal necrotic postreconstruction after 5 days of follow-up, which required a reexcision. Two patients were diagnosed with phyllodes breast tumor, and three other patients were diagnosed with breast malignancies. Two patients underwent breast tumor excision, while the other three were treated with radical mastectomy. All of these chest wall defects were closed using 120-degree skin flap reconstruction from the primary defect center circle, using half of the diameter of the skin defect for closure. All patients were also satisfied with the postreconstruction result, with minimal postoperative wound treatment.
Case 1
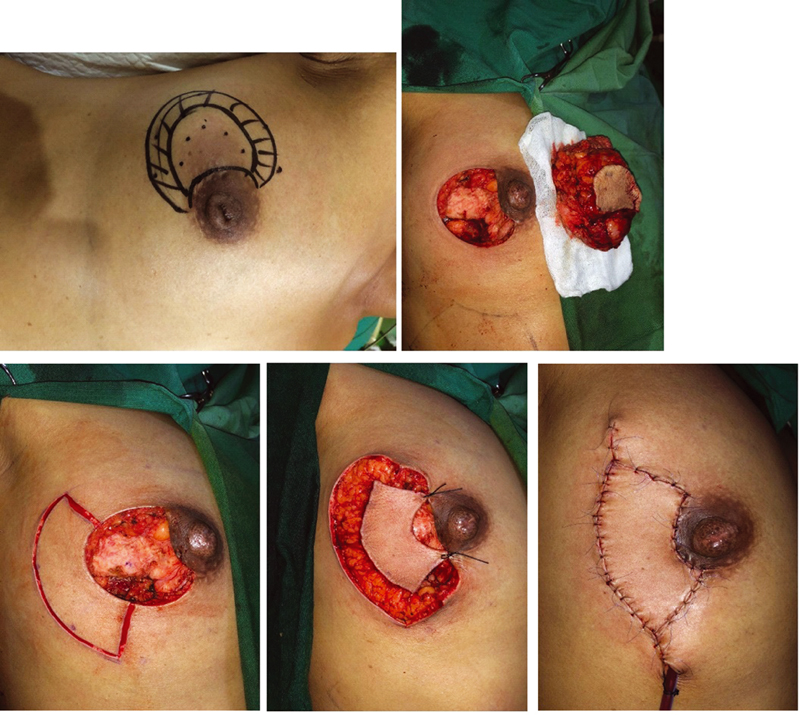
A 45-year-old patient (patient A) presented to our oncology surgery polyclinic with a solid mass in her right breast. She had already done an excision biopsy on her breast, and the histopathology result revealed a phyllodes tumor on her right breast. The patient did not feel any pain in her breast. Wide tumor excision was done, and a 4-cm postexcision defect was left. We used 120-degree angle for skin reconstruction, and 2-cm skin radius was needed for defect closure (Fig. 3). Chest defects were successfully closed within 2 hours of operative duration. The patient was discharged from the hospital after 4 days of treatment. No postoperative complications were observed from 1-month follow-up in our clinic.
-
Fig. 3 A 45-year-old patient with small phyllodes tumor on her right breast underwent horse-she flap reconstruction using 120-degree angle from the center circle and 2-cm defect radius.
Case 2
A 36-year-old patient (patient B) presented to our surgery polyclinic with a large mass located in the lower medial quadrant of her left breast. An excision biopsy was done before admission from another hospital, and the histopathology result showed a phyllodes tumor, similar to the previous patient but bigger in diameter size. She was offered a choice to remove her left breast entirely, including her solid mass from her previous hospital, but the patient refused and wished to preserve her left breast while removing the tumor. Thus, the patient was referred to our care and we decided to perform wide excision of the tumor and close the chest wall defect with horse-shoe flap reconstruction to preserve the left breast. Wide tumor excision was done, and a defect of 8 cm was left as a result. Using the 120-degree angle from defect center circle, 4-cm length of adjacent skin outside the primary defect was needed to close the primary defect itself. Primary tumor removal and chest reconstruction using horse-shoe flap technique were successfully done, completely removing the entire tumor and preserving the left breast with only a small section of breast subcutaneous fat tissue removed (Fig. 4). We continued our follow-up examination after the patient was discharged, and the result was satisfactory after 1 month of wound care.
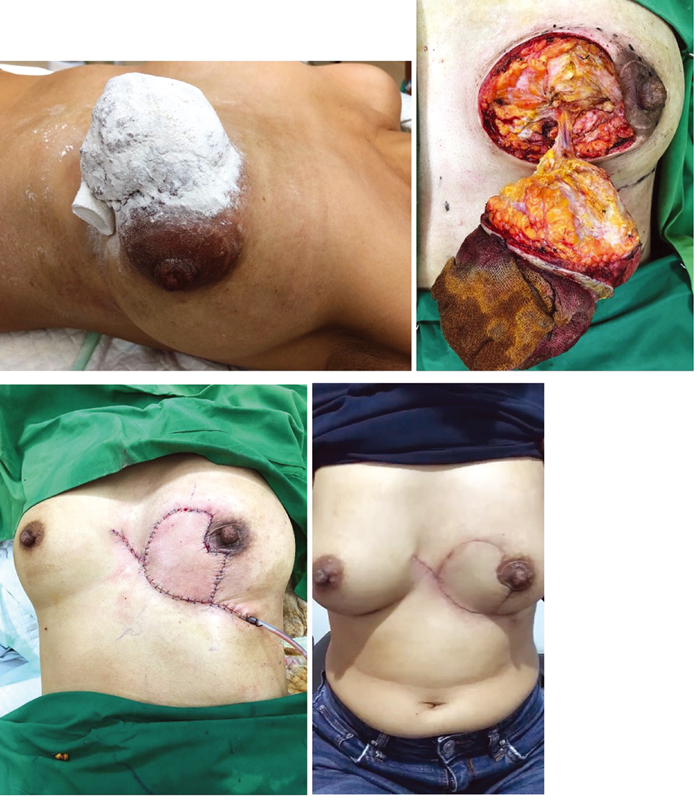
-
Fig. 4 A 36-year-old patient with a bigger phyllodes tumor underwent wide excision and reconstruction with horse-shoe flap, 120-degree angle from the center circle and 4-cm defect radius.
Case 3
A 50-year-old patient (patient C) was presented with multiple palpable masses in her right breast. She was diagnosed with invasive breast cancer, ductal carcinoma with triple-negative molecular type. Six series of neoadjuvant chemotherapy were done but did not seem to show any improvement clinically. A horse-shoe flap design was planned for reconstructing the chest wall after we dissected the remaining tumor on her right breast. We shaped and designed a circular defect with 11-cm diameter. This length was considered necessary and expected to leave a safe margin. With 11 cm of primary site defects and 120-degree angle from the center of circular defects, 5.5 cm of adjacent tissue outside the primary defect was sufficient to cover the defect. We removed all tissue from the circular area and preserved the pectoralis minor muscle. The reconstruction was successfully done, leaving with minor scar tissues (Fig. 5). The patient was followed-up until 1 month after discharge, and there were no surgical complications.
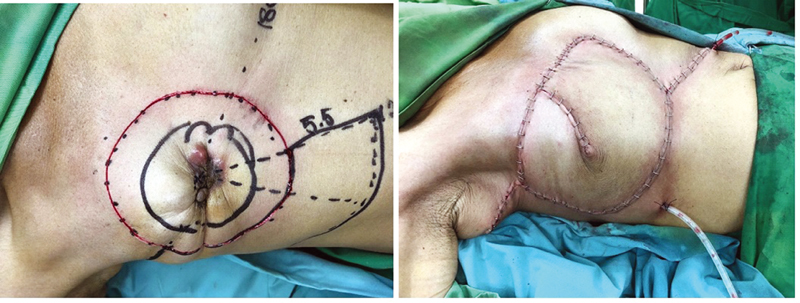
-
Fig. 5 A 55-year-old patient with invasive breast cancer underwent radical mastectomies with 11-cm diameter of primary defects, 5.5 cm of adjacent tissue radius was used to close the defects using a horse-shoe flap.
Case 4
A 52-year-old female (patient D) was our patient who routinely came to our clinic for regular monthly examination. She was diagnosed with T4N2M0 malignant breast cancer, ductal carcinoma, and triple-negative molecular type. Six series of neoadjuvant chemotherapy were delivered with no improvement and clinical response. A large solid mass dominated her left breast, with an open ulcer on her upper lateral quadrant. There were granulation tissues on the ulcer edges with infections, pus, and devitalized tissue on the center. Radical mastectomy was planned, with a horse-shoe flap technique for defect closure. We drew a circular pattern around the breast which measured around 21 × 17 cm2 of primary defect expected after the radical mastectomy. The entire primary tumor and breast tissues were removed with preserved pectoralis muscle. We used a 120-degree angle from the center circular defect, half of the diameter defect length (around 10 cm) of adjacent tissue inferior to the primary defect (Fig. 6). The patient was followed-up until 2 months after successful reconstruction. There were some serous fluid formations below the reconstruction site but the result was satisfactory after a few routine aspirations.
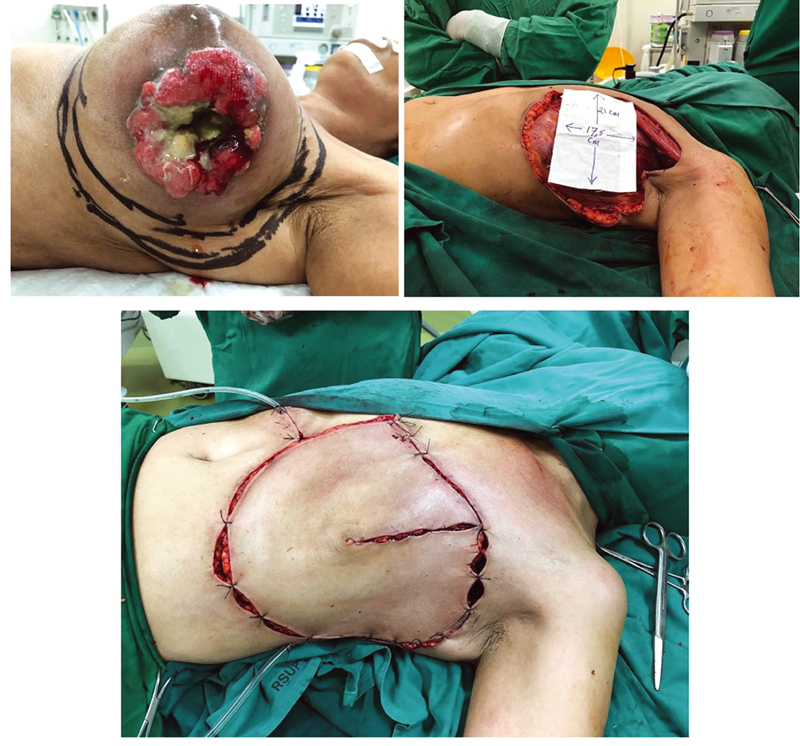
-
Fig. 6 A 52-year-old patient with 21 × 17 cm2 of the primary defect after radical mastectomy underwent a horse-shoe flap using 10 cm of skin outside the defect for closure.
Case 5
A 55-year-old female (patient E) with luminal-type T4N1M0 malignant breast cancer, presented with a large breast tumor on her left breast. Twelve series of neoadjuvant chemotherapy were given, with the aim to downsize the tumor, but no significant response was seen in postchemotherapy clinical evaluation. Radical mastectomy and horse-shoe flap technique were planned as operative approach and soft tissue coverage. A circular pattern was needed for horse-shoe flap coverage, and it was estimated around 26 cm of defect diameter size as a result to achieve safety tumor margin. Adjacent tissue of 13 cm below the primary defect was needed for chest wall coverage, as we used 120-degree angle from the center defect. There were no complications shortly after surgery. Five days after surgery, marginal necrotic skins were found around the interrupted surgical stitches and did not seem viable and irreversible. Reexcision of necrotic skin was required, and only small parts of necrotic skin were removed. After stitching both edges where necrotic skins were removed, the patient's follow-up examination was continued for 1 month, and there were no other complications afterward (Fig. 7).
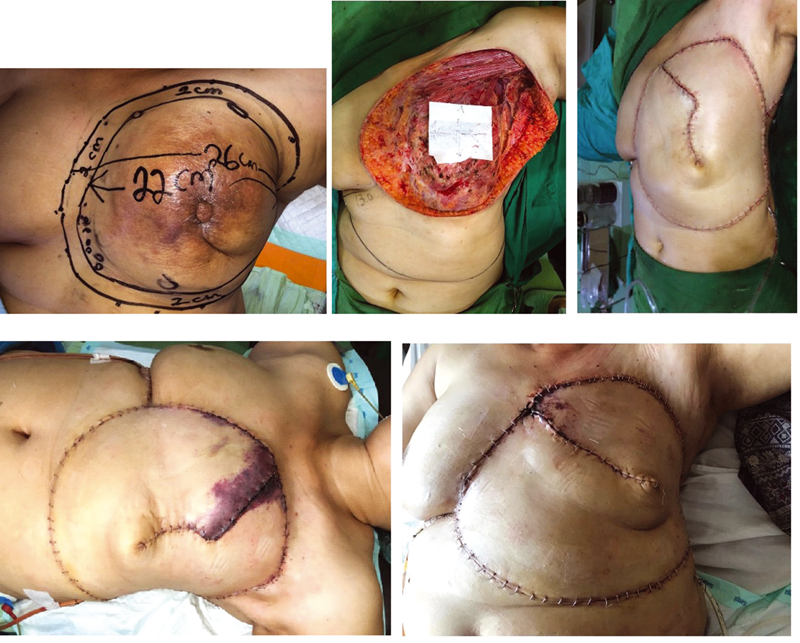
-
Fig. 7 A 55-year-old patient with the largest defect in this study underwent horse-shoe flap using 13-cm adjacent tissue below the defects. Marginal necrotic skins were found after surgery, and reexcision of necrotic skin was necessary, followed by stitching both edges for closure.
Discussion
In several cases of large breast tumors, extensive chest wall resections are needed to reassure free margins, thus leaving massive defects that require closure approaches. Several techniques have already been introduced; some are widely used, such as latissimus flap, abdominal flaps, and other donor sites, such as superior gluteal artery perforator flaps or using contralateral healthy breast for chest wall defects, that did not cross the midline. Chest wall closures are necessary for patients with large primary breast tumors, recurrent breast tumor with radiation therapy (some patients with radiation-induced angiosarcomas are indicative for wide skin resections), and in patients with inflammatory breast cancers. Closing the chest defects after wide tumor resections only using primary simple skin closure is sometimes insufficient, and additional techniques for chest wall closure are obligatory.6
The easiest way, but no longer used by many experts, for chest skin coverage is split-thickness skin grafts (STSG). STSGs are widely known and best used for relatively minor and small skin defects with sufficient vascular beds. However, STSG for chest wall closure takes longer to heal (in some cases more than 6 weeks), unstable skin coverage which left sloughing of the grafted skin, contracture of the skin graft, and other complications that may emerge, particularly after postclosure radiation therapy. This technique is not a suitable option for large chest wall defects.6 The horse-shoe flap technique proved that it could be applied from small defects into large defects with minor complications. The largest diameter which had already been tested was a 26-cm skin defect. We assured that this technique is applicable in variable sizes but further trials are needed along with defining indications and contraindications in utilizing the horse-shoe flap.
LD muscle flap is one of the most frequent techniques used for chest wall coverages. The LD muscle is the largest muscle located throughout the chest area.3 LD flap is often used as a choice for autologous breast reconstruction and to salvage previous failed breast reconstruction. LD can be safely applied for patients who underwent radiation therapy prior to breast conservation therapy. LD can also be applied for multiple breast sizes which can leave various size defects post-mastectomy. Some LD flaps can cause complications; one of the most common is seroma formation, particularly in patients with extended LD flaps. Other common complications include wound separations and dehiscence, skin flap necrosis, shoulder weakness, restricted affected arm movements, and dorsal hernia.7 The horse-shoe flap reconstruction can facilitate patients with LD flap, particularly patients with large primary defects, to completely cover all resected areas. Combined with LD flaps, all defects from large radical mastectomies or even secondary defects, taken from extended LD reconstruction can be safely covered. Further trials and reports for these combined reconstruction techniques are needed.
For almost 40 years, pedicled transverse rectus abdominis musculocutaneous flaps have been widely used and performed in breast cancer patients, routinely performed by plastic surgeons and oncologists worldwide. This autologous chest wall reconstruction can be offered to patients with a body mass index below 30 kg/m2 and with enough adipose tissue on their abdominal area.7 It can be divided into two techniques, pedicled transverse rectus abdominis musculocutaneous (TRAM) flap which maintains superior epigastric vessels as its vascular supply or free TRAM flap. This TRAM technique leaves an additional scar in the abdominal area while also requiring longer operating time and hospitalization. Partial flap necrosis, breast asymmetry, and hernia are the most common complications found from this technique.8 One of the complications mentioned above from horse-shoe flap is also partial flap necrosis, but it can be easily managed by removing it partially by removing the necrotic edges and stitching all the edges back with a simple suture. For the next 30 days, the flap was still viable after necrotic skin removal. This is due to the fact that horse-shoe flap has multiple perforator vessels, bringing more vascular supply needed for flap safety and larger area coverage.
Some authors mentioned about the development of local recurrence following mastectomy and autologous breast reconstructions in patients with malignant breast cancer. One observation study by Wu et al revealed 11 patients (of 397 samples) with local recurrence after autologous breast reconstruction, with most of them were reconstructed using LD flap. The median time needed for local recurrence to develop was 2.9 years.9 Other retrospective study by Mirzabeigi et al also reported a total of 41 patients with locoregional recurrence after mastectomies and breast reconstructions, 13 of them used implant reconstruction, and 17 patients underwent autologous breast reconstructions.10 This particular topic is the main focus of the study, as we have not done any survival analysis in our patients with horse-shoe flap. Although other risk factors contributed to locoregional recurrence events, we would like to investigate whether this reconstruction technique can be one of the protective factors related to locoregional recurrences.
One of the newest breakthrough methods, which is widely and frequently used, is adjacent tissue coverage where local tissues are rearranged as flaps to cover skin defects as an alternative to skin grafts and free flaps.6 It can be used in patients with larger defects and is much simpler than mentioned above. The most common adjacent tissue flap and the most famous is the keystone flap, widely used in dermatologic surgery. It relies on musculocutaneous and vascular perforators and flaps base, increasing its reliability, as blood flow is the key factor for flap's tenacity.11 Keystone flap, as one of the adjacent tissue coverage, has many advantages. It can be applied in almost every body region, has a simple design, and is easy to master.12 Similar to the keystone flap, the horse-shoe reconstructive technique also has a simple design and is easy to perform. We have not tried this flap technique for defects in other body regions. Further study and trials are needed to verify its application for other defect closure. Visually, the major difference between the keystone island flap and our horse-shoe flap is the design. Keystone flap can be applied by creating an elliptical shape defect, while the horse-shoe flap is designed from a circular shape. Both designs are easy to create but theoretically, circular shape defects are safer since it removes more adequate tumor margins than elliptical shape defects. Based on the author's knowledge, there are no case reports regarding the usage of keystone flaps for post-mastectomy chest skin defects or breast-conserving therapy.
One interesting advantage of this circular shape adjacent flap reconstruction is the availability of surrounding healthy tissue that can be taken as a flap. Keystone flap only relies on the skin under the elliptical incision, and the flaps below will be advanced upward to close the defect. The horse-shoe flap offers variable options, where it can use all of the surrounding tissues, depending on the defect's diameter and angle from the center circle. Based on these factors, the adjacent flap can be taken from under the incision and can be taken from any direction around the defects. Based on the author's experience and knowledge, there is not any reconstruction technique, especially the adjacent flap technique which provides every angle of surrounding skin as an available flap to date.
Conclusion
Our novel horse-shoe flap technique proved to be an excellent option for closing chest skin defects. It can be applied for variable defect sizes with minor complications. The horse-shoe flap, as mentioned above, has multiple perforators, leading to better flap viability and a broader coverage area. As one of the adjacent tissue coverage, this reconstruction technique is easy to apply and perform. It offers freedom to every surgeon, as this technique can provide all surrounding skin to be a usable flap. This simple method of breast reconstruction breaks every boundary and limitation that surgeons have faced before. It opens new paths regarding safer and fewer complications breast reconstruction. Several trials are needed to evaluate the strengths and weaknesses of our flap technique. Further study regarding this technique will be expected in the future.
Conflict of Interest
None declared.
References
- Chest wall resections and reconstruction: a 25-year experience. Ann Thorac Surg. 2002;73(06):1720-1725. , discussion 1725–1726
- [Google Scholar]
- Review of procedures for reconstruction of soft tissue chest wall defects following advanced breast malignancies. Cancer Contr. 2019;26(01):1073274819827284.
- [Google Scholar]
- Chest wall reconstruction without prosthetic material. Thorac Surg Clin. 2017;27(02):165-169.
- [Google Scholar]
- Superior epigastric artery perforator flap: anatomy, clinical applications, and review of literature. J Reconstr Microsurg. 2014;30(07):475-482.
- [Google Scholar]
- Reciprocal incisions for closure of circular skin defects. Plast Reconstr Surg. 1981;67(04):482-491.
- [Google Scholar]
- Breast reconstruction In: Harris J R, Lippman M E, Morrow M, Osborne C K, eds. Diseases of the breast. 5th ed. Philadelphia, PA: Wolters Kluwer Health; 2014. p. :536-549.
- [Google Scholar]
- Breast reconstruction with the pedicled transverse rectus abdominis musculocutaneous (TRAM) flap In: Pu L Q, ed. Karp N S, ed. Atlas of Reconstructive Breast Surgery. Edinburgh, Scitland: Elsevier; 2020. p. :1-15.
- [Google Scholar]
- Autologous reconstruction TRAM flap In: Bland K I, ed. Klimberg V S, ed. Master Techniques in General Surgery: Breast Surgery. Philadelphia, PA: Wolters Kluwer Health; 2011. p. :439-441.
- [Google Scholar]
- Local recurrence following mastectomy and autologous breast reconstruction: incidence, risk factors, and management. OncoTargets Ther. 2016;9:6829-6834.
- [Google Scholar]
- Locoregional cancer recurrence after breast reconstruction: detection, management and secondary reconstructive strategies. Plast Reconstr Surg. 2019;143(05):1322-1330.
- [Google Scholar]
- Basic principles in flap reconstruction In: Flaps and Grafts in Dermatologic Surgery. 2nd ed. Edinburgh, Scotland: Elsevier; 2019. p. :16-17.
- [Google Scholar]
- Design principles and the keystone technique In: The Keystone Perforator Island Flap Concept. Sydney, Australia: Elsevier; 2012.
- [Google Scholar]







